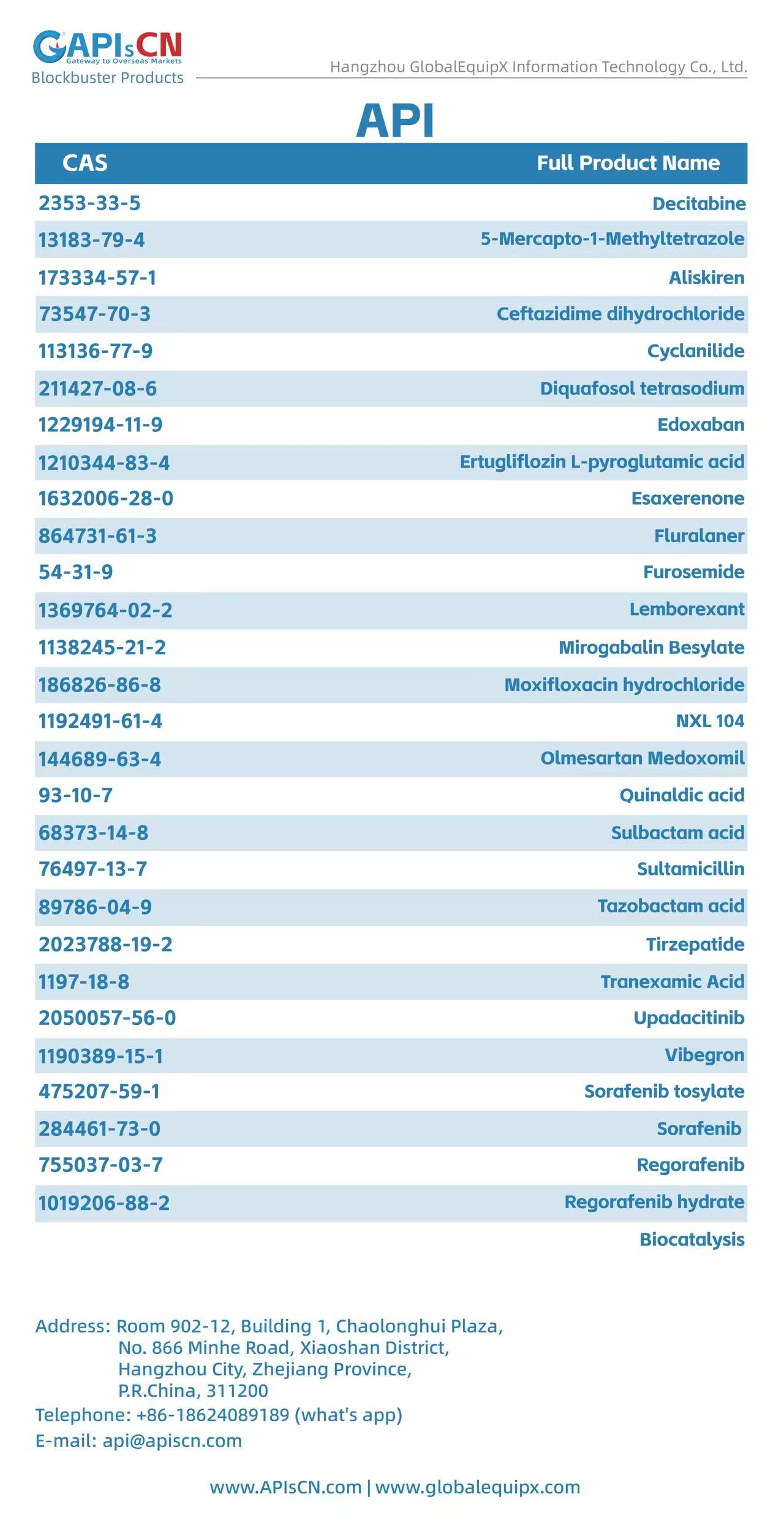
Moxifloxacin Hydrochloride (CAS: 186826-86-8) is a GMP-compliant, high-purity 4th-generation fluoroquinolone antibacterial API. It delivers broad-spectrum bactericidal efficacy via inhibiting bacterial DNA gyrase and topoisomerase IV, widely used for respiratory, intra-abdominal, skin and ophthalmic infections. It fully meets USP, EP, CP and ICH standards for oral, injectable and ophthalmic formulation production.
Olmesartan Medoxomil (CAS 144689-63-4) is a high-purity, GMP-grade angiotensin II receptor antagonist (ARB) API. It is indicated for the treatment of hypertension, providing long-lasting blood pressure control with good tolerability. It meets USP, EP, and ICH quality standards, suitable for pharmaceutical formulation development and commercial production.
Tazobactam Acid (CAS 89786-04-9) is a GMP-compliant, high-purity β-lactamase inhibitor active pharmaceutical ingredient (API). It irreversibly inactivates bacterial β-lactamases, widely used in combination with β-lactam antibiotics to combat drug-resistant gram-negative bacterial infections. It fully meets USP, EP and ICH international quality standards for pharmaceutical formulation manufacturing
Poly-L-Lactic Acid (Microsphere) CAS: 33135-50-1
Polycaprolactone (Microsphere) CAS: 24980-41-4
Our hours
Monday to Friday
(8:30-17:30)